MYCORRHIZAL ASSOCIATIONS: The Web Resource
Section 4. ARBUSCULAR MYCORRHIZAS
A. Introduction
Mycorrhizal associations produced by Glomeromycotan fungi are known as arbuscular mycorrhizas, or vesicular-arbuscular mycorrhizas (formerly also endomycorrhizas, or endotrophic mycorrhizas) and are abbreviated as VAM here. There is disagreement about whether arbuscular mycorrhizas or vesicular-arbuscular mycorrhizas is the most appropriate name to, because some fungi do not produce vesicles, but arbuscules are not consistently used to identify associations (i.e. they are absent in myco-heterotrophs and older roots) (see Section 10). These associations involve primitive fungi in the Glomeromycota and a wide diversity of plants, as explained at the end of this Section. Components of associations are illustrated below with details in the following sub-sections.
| Topic | Major Sources |
| Images | Brundrett et al. 1985, Brundrett & Kendrick 1988, Brundrett et al. 1996 |
| Diagrams and definitions | Brundrett et al. 1996 |
| Glomeromycotan fungi | Arthur Schüßler's site |
 |
A. Structures in Soil
B. Structures in Roots
|
B. Looking at Associations
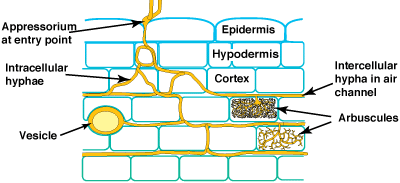
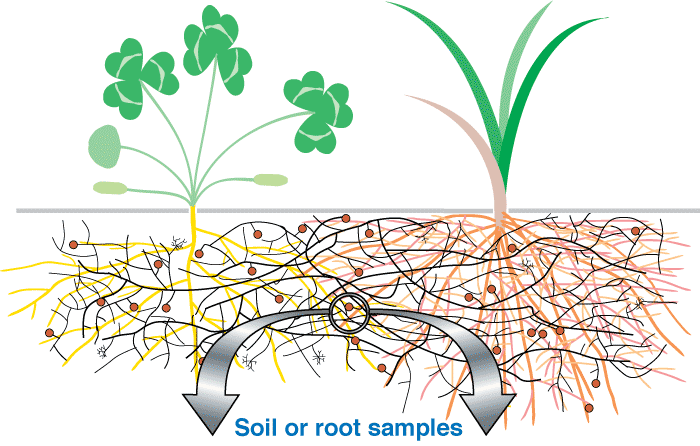
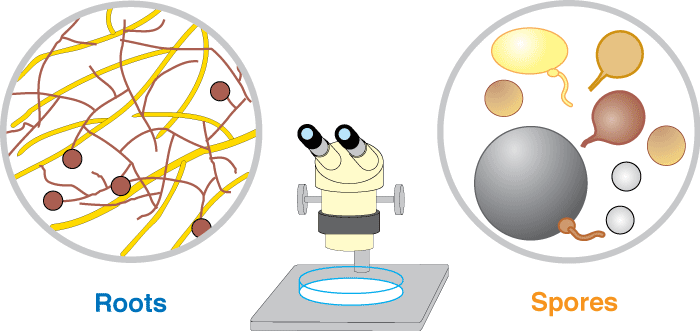
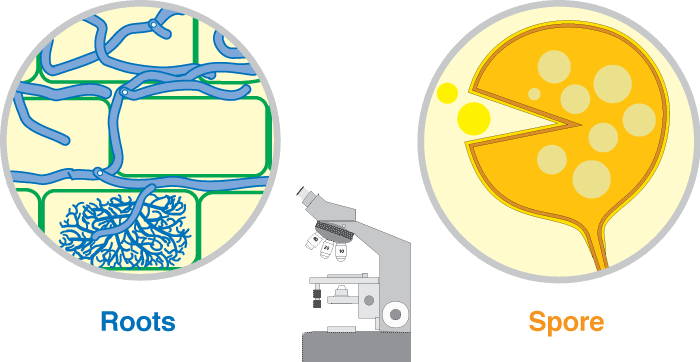
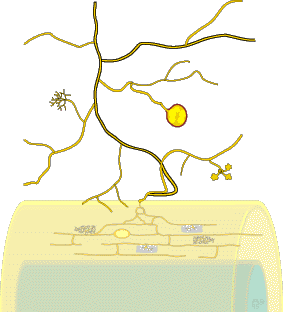
These associations are introduced by 3 diagrams showing increasing magnification views. Methods used to see mycorrhizal structures are described in Section 10.
Diagrams on this page are also available as pdf files to download.
Level 1. Whole Plants
 |
|
|
Level 2. The Dissecting Microscope
 |
|
|
Level 3. The Compound Microscope
 |
|
|
|
C. Structures and Developmental Stages
VAM associations form when host roots and compatible fungi are both active in close proximity and soil conditions are favourable. The stages in root colonization by VAM fungi are illustrated below. Associations formed by species of Glomus, Gigaspora, Scutellospora and Acaulospora are shown. Terminology is explained in the glossary.
1. Soil Hyphae
Mycorrhizal associations may be initiated by spore germination as illustrated here. Hyphae may also originate from fragments of roots. In many cases there already is a pre-existing network of hyphae resulting from previous root activity. Hyphae resulting from spore germination have a limited capacity to grow and will die if they do not encounter a susceptible root within a week or so. Hyphae emerge from a germination shield within the spore in Scutellospora and Acaulospora species.
 |
Germinating hyphae which emerged several days after spores were extracted from dry soil. These spores are of Gigaspora decipiens (left) and Scutellospora cerradensis (right).
|
 |
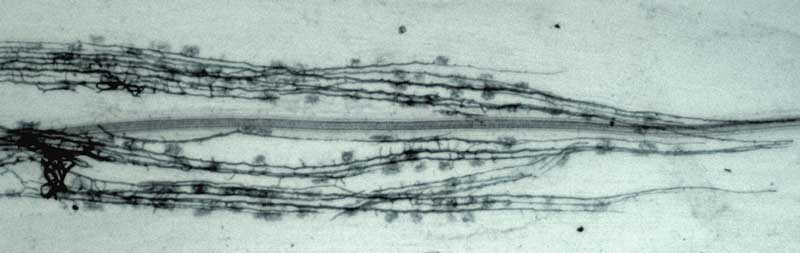
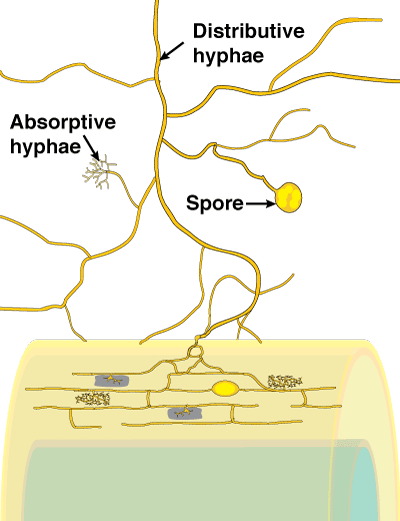
Soil hyphae, also known as extraradical or external hyphae, are filamentous fungal structures which ramify through the soil. They are responsible for nutrient acquisition, propagation of the association, spore formation, etc. VAM fungi produce different types of soil hyphae including thick "runner" or "distributive" hyphae as well as thin "absorptive" hyphae (Friese & Allen 1991). The finer hyphae can produce "branched absorptive structures" (BAS) where fine hyphae proliferate (Bago et al. 1998). Hyphae of Scutellospora and Gigaspora species produce clustered swellings with spines or knobs called auxiliary cells. Roles of hyphae are discussed in another section. The network of hyphae in the soil is only connected to roots by the entry points that initiate mycorrhizal associations, as hyphae do not grow out of living roots. |
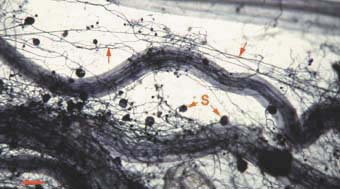 |
Mycorrhizal root system washed carefully from coarse sand to reveal the intact network with external hyphae (arrow) with spores (S) produced by Glomus mosseae.
|
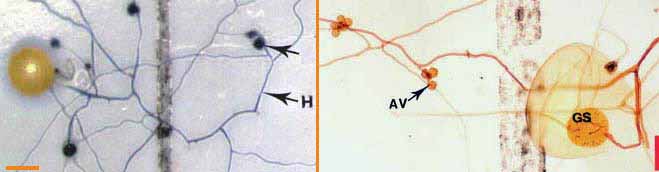
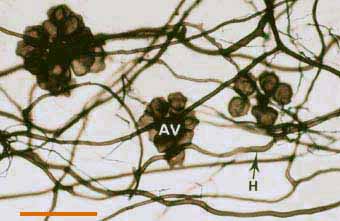 |

Darkly pigmented soil hyphae (H) of a Scutellospora species with auxiliary cells (AV).
|
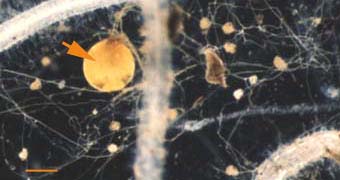 |
Soil hyphae produced by a single germinated spore of Gigaspora (arrow) used to start a mycorrhizal association. The hyphal network has produced accessory vesicles and is spreading through the root system of a clover plant.
|
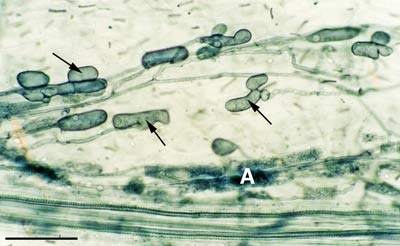
2. Root Contact and Penetration
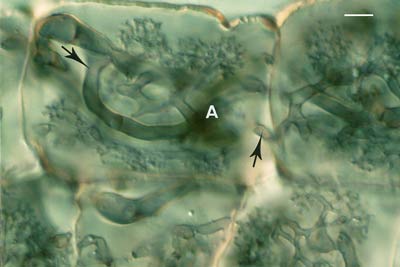
Mycorrhizal associations start when soil hyphae respond to the presence of a root by growing towards it, establishing contact and growing along its surface. Next, one or more hyphae produce swellings called appressoria between epidermal cells. Root penetration occurs when hyphae from the appressoria penetrate epidermal or cortical cells to enter the root. These hyphae cross the hypodermis (through passage cells if these are present in an exodermis) and start branching in the outer cortex. Relevant root structural features are explained in Section 2.
The images in the following sections are of cleared roots which have been stained with Chlorazol black E as explained in Section 10. Many images are from a study of the time course of VAM formation by Glomus versiforme in leek (Allium porrum) roots (Brundrett et al. 1985). | |
 |
Soil hyphae have produced 2 appressoria between epidermal cells (arrows). These are seen here in a surface view of a root with attached hyphae.
|
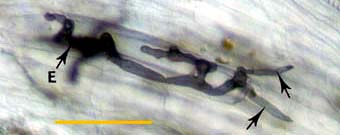 |
Hyphae at an entry point (E) penetrating cortex cells (arrows) approximately 1 day after contact with the root.
|
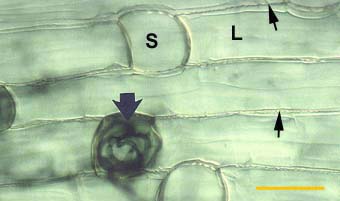 |
Alternating long (L) and short (S) cells in the dimorphic exodermis of a Smilacina racemosa root. Hyphae of VAM fungi have penetrated unsuberised short cells (arrows).
|
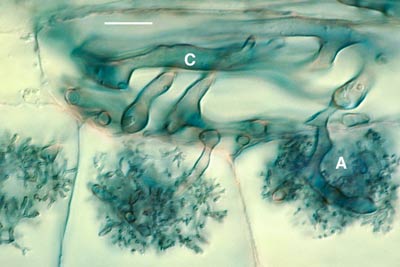
3. Hyphal Proliferation in the Cortex
Aseptate hyphae spread along the cortex in both directions from the entry point to form a colony. Hyphae within root are initially without cross walls, but these may occur in older roots. Gallaud (1905) observed that VAM associations in different species formed two distinctive morphology types, which he named the Arum and Paris series after host plants. These are now known as linear (1.1) and coiling (1.2) associations respectively, as explained in Section 1. The differences between these two modes of spread within the root cortex are illustrated below. Both types of associations are important in ecosystems (Smith & Smith 1997).
- Linear (Arum) series associations where hyphae proliferate in the cortex by growing longitudinally between host cells. This occurs because hyphae grow through longitudinal intercellular air spaces that are present (Brundrett 2004).
- Coiling (Paris) series where hyphae spread by forming coils within cells because there are no continuous longitudinal air spaces.
(i) Coiling (Paris) Arbuscular Mycorrhizas
These are associations where hyphae spread primarily by intracellular growth following a convoluted path through cortex cells. The resulting colonies of VAM fungi generally have a coiled appearance (Gallaud 1905, Brundrett & Kendrick 1998), but may have more digitate branching patterns (Widden 1996). Arbuscules may be restricted to a single layer of cells in the inner cortex.
(ii) Linear (Arum) Arbuscular Mycorrhizas
These are associations where hyphae grow along longitudinal intercellular air channels between the walls of root cells. A relatively rapid parallel spread of intercellular hyphae may occur along these channels. The resulting colonies of VAM fungi have a linear appearance.
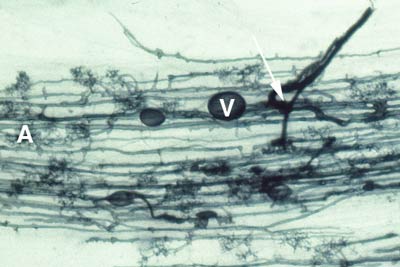
4. Arbuscules
Arbuscules are intricately branched haustoria that formed within a root cortex cell. They were named by Gallaud (1905), because they look like little trees. Arbuscules are formed by repeated dichotomous branching and reductions in hyphal width, starting from an initial trunk hypha (5-10 um in diameter) and ending in a proliferation of fine branch hyphae (< 1 um diameter).
Arbuscules start to form approximately 2 days after root penetration (Brundrett et al. 1985). They grow inside individual cells of the root cortex, but remain outside their cytoplasm, due to invagination of the plasma membrane. Arbuscules are considered the major site of exchange between the fungus and host. This assumption is based on the large surface area of the arbuscular interface, but has not been confirmed (Smith 1995). Arbuscule formation follows hyphal growth, progressing outwards from the entry point. Arbuscules are short-loved and begin to collapse after a few days, but hyphae and vesicles can remain in roots for months or years.
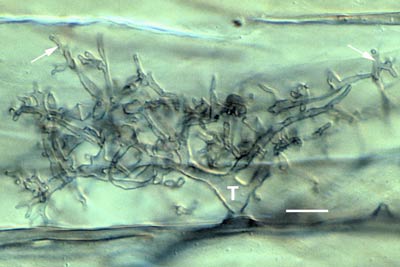 |
Developing arbuscule of Glomus mosseae in a root cell with fine branch hyphae (arrows). The trunk (T) of this arbuscule branched from an intercellular hyphae.
|
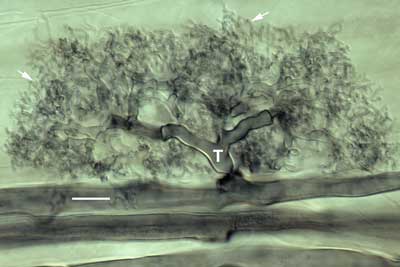 |
Mature arbuscule of Glomus showing trunk (T) and numerous fine branch hyphae (arrows). Zoom in with roll-over
|
 |
An arbuscule of Glomus versiforme in a root cortex cell with branch hyphae densely packed in the cortex cell of the host.
|
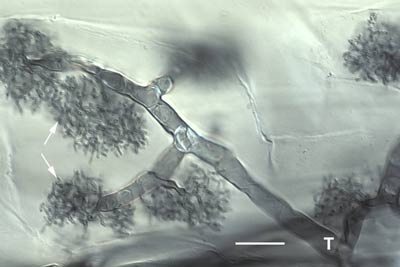 |
Arbuscule of Gigaspora margarita with an elongated trunk hypha (T) and tufts of fine branch hyphae (arrows). Note how this arbuscule differs from the Glomus arbuscules above.
|
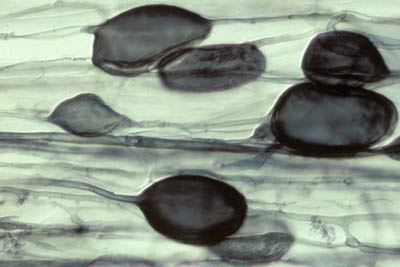
5. Vesicles
Vesicles develop to accumulate storage products in many VAM associations. Vesicles are initiated soon after the first arbuscules, but continue to develop when the arbuscules senesce. Vesicles are hyphal swellings in the root cortex that contain lipids and cytoplasm. These may be inter- or intracellular. Vesicles can develop thick walls in older roots and may function as propagules (Biermann & Linderman 1983). Some fungi produce vesicles which are similar in structure to the spores they produced in soil, but in other cases they are different.
6. Structural Diversity
It is possible to identify individual Glomeromycotan fungi by recognising characteristic root morphology patterns in roots (Abbott 1982). Identification of endophytes within roots is important for culture quality control, because contaminating fungi can be identified months before they sporulate (Brundrett et al. 1999). This procedure can also be used to determine the mycorrhizal inoculum potential of different fungi by growing trap plants in a soil.
It is also possible to identify Glomeromycotan fungi by colonization patterns in roots, but it is difficult to separate species. Morphological features that are important include variations in vesicles (size, shape, wall thickness, wall layers, position and abundance), hyphal branching patterns, the diameter and structure of hyphae (especially near entry points), and the staining intensity of hyphae (dark or faint). Characteristics of genera of Glomeromycotan fungi are listed and illustrated below (or downoad diagram).
| Mycorrhizas produced by Glomus species: | |
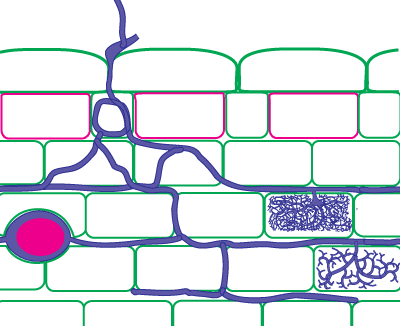 |
|
| Mycorrhizas produced by Scutellospora and Gigaspora species: | |
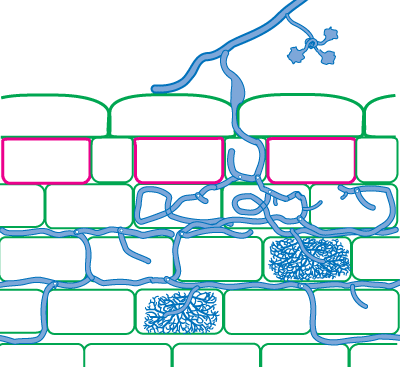 |
|
| Mycorrhizas produced by Acaulospora species: | |
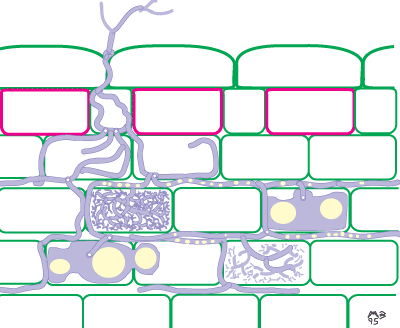 |
|
| Mycorrhizas produced by fine endophytes: | |
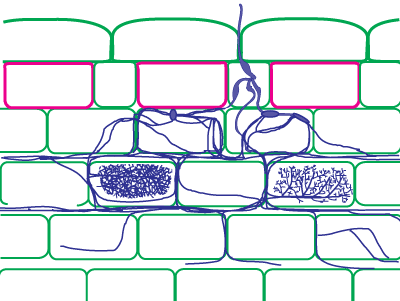 |
|
7. Spores
Spores form as swellings on one or more subtending hypha in the soil or in roots. These structures contain lipids, cytoplasm and many nuclei. Spores usually develop thick walls with more than one layer and can function as propagules. Spores may be aggregated into groups called sporocarps. Sporocarps may contain specialised hyphae and can be encased in an outer layer (peridium). Spores apparently form when nutrients are remobilised from roots where associations are senescing. They function as storage structures, resting stages and propagules. Spores may form specialised germination structures, or hyphae may emerge through the subtending hyphae or grow directly through the wall.
Diversity of spores in a single soil sample
| |
 |
 |
Left: Spores separated from soil and sorted into categories based on size and colour. Right: Spores (S) on a piece of filter paper used to start a "pot culture" using pasteurised soil in which a host plant was grown . | |
The images below show some of these spores viewed with a compound microscope. Inner wall layers have been revealed by crushing spores and by staining with Melzer's reagent (I2KI). All scale bars = 100 µm. | |
| Spores of Glomus | |||
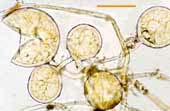 |
Relatively small white spores of a Glomus species. |
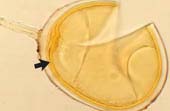 |
Spore of Glomus clarum which has a visible inner wall layer (arrow). |
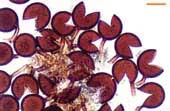 |
Sporocarp of Glomus invermaium typical of the dead spores often found in field-collected soil. |
 |
Living spores of Glomus invermaium from a pot culture. |
| Spores of Acaulospora | |||
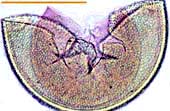 |
Acaulospora spore with deep pits in the outer wall and inner wall layers stained by Melzer's reagent. |
 |
Acaulospora spore with several inner wall layers (arrows). One layer has stained darkly with Melzer's reagent. |
| Spores of Scutellospora Species | |||
 |
White Scutellospora cerradensis spores with prominent brown germination shields. |
 |
Large black spore with deep pits of Scutellospora reticulata. |
D. Arbuscular Mycorrhizal Fungi
Populations of arbuscular mycorrhizal fungi in the Glomeromycota are thought to have occupied the same soil habitats for millions of years, slowly adapting to changes in site conditions (Trappe & Molina 1986, Brundrett 2002). Many of these fungi have worldwide distribution patterns, but soil factors such as pH restrict the distribution of other taxa (Abbott & Robson 1991). Consequently, habitat information is as important as knowledge of the taxonomic identity of fungi, for comparing the results of experiments, or the selection of isolates for practical use.
The classification of the Glomeromycota is based on the structure of their soil-borne spores and DNA sequences. Accurate identification of these fungi often requires them to be isolated in cultures with host plants, to observe developmental stages, avoid the loss of diagnostic features and obtain healthy spores for DNA extraction. There now are many DNA sequences of Glomeromycotan fungi in databases such as Genbank. Information on Glomeromycotan systematics is available via the links below.
Arbuscular Mycorrhizal Fungi
- VAM fungi belong to the Glomeromycota.
- They are primitive fungi at the base of the tree for higher fungi (basidiomycetes).
- They associated with first land plants and appear to have evolved very slowly since then.
- They have no known sexual state.
- They produce microscopic structures, or relatively small sporocarps (truffle-like).
- Just over 200 species of these fungi are described, yet they are capable of forming mycorrhizal associations with the majority of plants.
Classification scheme for Glomeromycotan taxa
This classification follows Schüßler et al. (2001) with emendations of Oehl and Sieverding (2004), Walker and Schüßler (2004), Sieverding and Oehl (2006), and Spain et al. (2006). Visit Arthur Schüßler's site for further information and a family tree for Glomeromycotan fungi.
| Taxonomic Hierarchy | List of genera | ||||||||||||||||||||||
|
|
Links
- International Culture Collection Of Arbuscular and Vesicular-Arbuscular Mycorrhizal Fungi (INVAM)
- Glomeromycota in vitro collection (GINCO)
- Banque Europeene de Glomales (BEG)
- Arthur Schüßler's site
- Janusz Blaszkowki's website
- Glomeromycota on the Tree of life site
E. Host Plants
Plant families which are nonmycorrhizal or have ectomycorrhizas are listed in the relevant sections of this website and it is expected that that the majority of other plant families have VAM associations. Families of Australian plants with VAM are listed in Section 8.
F. Terminology
This glossary defines some important terminology used to describe arbuscular mycorrhizas.
- Appressoria
- Hyphal swellings between two adjacent root epidermal cells. These are sites where hyphae first penetrate root cells by exerting pressure and/or enzymatic activity.
- Arbuscules
- These are intricately branched "haustoria" that form within root cortex cells that look like little trees (Gallaud 1905). Arbuscules are formed by repeated dichotomous branching and reductions in hyphal width from an initial trunk hypha that ends in a proliferation of very fine branch hyphae. They are considered to be the major site of exchange with the host plant. Old arbuscules collapse progressively until only the trunk remains. Collapsed arbuscules are sometimes called peletons.
- Aseptate hyphae
- These are hyphae which are without cross walls (coenocytic hyphae). Cross walls may form as hyphae age.
- Auxiliary bodies
- These structures, which are also called external vesicles /or accessory bodies, are clustered swellings on external hyphae. These are often ornamented by spines or knobs and are characteristic of Scutellospora and Gigaspora. These apparently do not function as propagules.
- Colony
- Hyphal colonization of a root resulting from one external hypha (these may arise from several adjacent entry points). These are often called infection units.
- Dichotomous branching
- A symmetrical branching pattern which occurs when two branches arise simultaneously from the tip of a hyphae, plant or fungus organ. Divergent branches grow at the same rate.
- Internal hyphae, intraradical hyphae
- Hyphae which grow within the cortex of a root to form a colony and later develop arbuscules and vesicles. These comprise the body (thallus) of a fungus in the root.
- Intercellular hyphae
- Hyphae which grow between the walls of adjacent root cells. These are in the root apoplast -- the zone outside the cytoplasm of cells.
- Intracellular hyphae
- Hyphae which grow within root cells. These penetrate the walls of cells and grow within them, but are separated from the cytoplasm by the plasma membrane.
- Soil hyphae
- These are also known as extraradical or external hyphae and are the filamentous structures which comprise the fungal thallus (body) in the soil. They acquire nutrients, propagate the association, produce spores and other structures. VAM fungi produce thick "runner" or "distributive" hyphae as well as thin, highly branched "absorptive" hyphae.
- Spores
- These are swollen structures with one or more subtending hyphae that form in the soil or in roots. Spores usually develop thick walls, which often have more than one layer. They can function as propagules. Spores of VAM fungi are sometimes called chlamydospores or azygospores.
- Sporocarps
- Aggregations of spores into groups, which may contain specialised hyphae and can be encased in an outer layer (peridium). Soil particles may be included in the spore mass. This term can be misleading, as the sporocarps produced by most Glomeromycotan fungi are small and relatively unorganised structures compared to those produced by larger fungi.
- Vesicles
- Intercalary (-o-) or terminal (-o) hyphal swellings formed on internal hyphae within the root cortex. These may form within or between cells. Vesicles accumulate lipids and may develop thick wall layers in older roots. The production and structure of vesicles varies between different genera of Glomeromycotan fungi. They are storage organs which may also function as propagules.
Version 2 © Mark Brundrett 2008